14 Reasons why Inhibikase Therapeutics, Inc. (Nasdaq: IKT) Could Be Poised For Significant Upside Potential in Early 2023.
Let’s Take a Closer Look at Inhibikase Therapeutics, Inc. (Nasdaq: IKT)
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) is a clinical-stage pharmaceutical company developing therapeutics for Parkinson’s disease, or PD, and related disorders.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) is a clinical-stage pharmaceutical company developing therapeutics for Parkinson’s disease, or PD, and related disorders.
The company’s multi-therapeutic pipeline has a primary focus on neurodegeneration and their lead program IkT-148009, an Abelson Tyrosine Kinase (c-Abl) inhibitor, targets the treatment of Parkinson’s disease inside and outside the brain as well as other diseases that arise from Abelson Tyrosine Kinases.
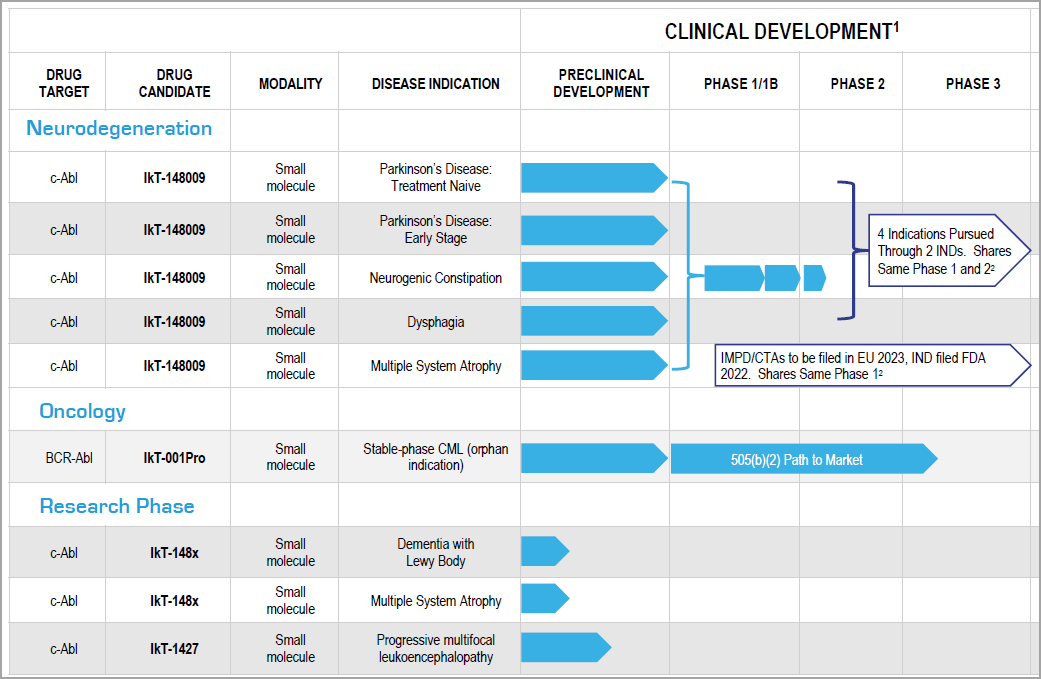
In 2021, Inhibikase Therapeutics, Inc. (Nasdaq: IKT) commenced clinical development of IkT-148009, a small molecule Abelson Tyrosine Kinase inhibitor which the company believes can modify the course of Parkinson’s disease including its manifestation in the gastrointestinal tract, or GI.
Results to date of the company’s completed Phase 1/1b Single and Multiple Ascending Dose escalation study (SAD and MAD, respectively) in older and elderly healthy volunteers and in mild to moderately advanced Parkinson’s patients have revealed important insights into the safety, tolerability, and pharmacokinetics of IkT-148009 in human subjects and patients.
Results from the 88 older and elderly healthy Phase 1 subjects and 14 Phase 1b Parkinson’s patients have shown that IkT-148009 has a half-life of greater than 24 hours and just a 25 mg once-daily oral dose reached exposures that are consistent with the exposures to the drug that resulted in therapeutic efficacy in animal models of progressive Parkinson’s disease.
In addition, a review of unblinded adverse event data revealed that just nine adverse events of any grade were observed in subjects or patients on IkT-148009, and at least four of these adverse events could not be attributed to IkT-148009.
The remaining five adverse events were of Grade 1 with no clinical significance.
FDA review of the Phase 1/1b data and the protocol for the Phase 2a three-month dosing study resulted in the FDA agreeing with the company’s view that it was appropriate for the Phase 2a study to begin, prompting them to close the Phase 1b study after two dosing cohorts.
The Phase 2a ‘201′ study began May 23, 2022 with the opening of the first site; the company has opened 16 of 34 selected sites as of November 1, 2022 and 11 patients have been randomized into the trial as of November 1, 2022.
120 treatment naïve patients are planned to be enrolled in this study which will dose patients with one of three planned doses of IkT-148009 or a placebo once daily for three months.
In addition to primary endpoints of safety/tolerability/pharmacokinetics, a hierarchy of 15 secondary endpoints measuring drug impact on motor and non-motor features of Parkinson’s disease in the brain or GI tract will be evaluated with descriptive statistics.
In July 2022, Inhibikase Therapeutics, Inc. (Nasdaq: IKT) filed its Investigational New Drug Application (IND) with the FDA in preparation to initiate the clinical development of IkT-001Pro, its prodrug of imatinib mesylate to treat Stable-phase Chronic Myelogenous Leukemia (SP-CML).
IkT-001Pro will be evaluated in a two-part dose finding/dose equivalence study in up to 56 healthy volunteers.
The study is designed to evaluate the steady-state pharmacokinetics of IkT-001Pro and determine the dose of IkT-001Pro equivalent to 400 mg imatinib mesylate, the standard-of-care dose for SP-CML.
Following clearance of the IND by the FDA on August 26, 2022, the two-part study, now known as the ‘501’ study, was initiated with the first patient dosing anticipated by the beginning of December, completion of the first cohort anticipated by year-end 2022 and completion of the pharmacokinetic analysis anticipated in early first quarter 2023.
Following the completion of both parts of this study and assuming the equivalent dose of IkT-001Pro relative to 400 mg imatinib mesylate is established, Inhibikase Therapeutics, Inc. (Nasdaq: IKT) will confer with the FDA to begin the NDA process following the proposed approval path for IkT-001Pro under the 505(b)(2) statute.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) will simultaneously pursue a superiority study comparing the selected doses of IkT-001Pro to standard-of-care 400 mg imatinib mesylate in SP-CML patients using a novel two-period wait list crossover switching study.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT)’s programs utilize small molecule, oral protein kinase inhibitors to treat neurodegenerative diseases and cancer.
In Parkinson’s disease (PD), Inhibikase Therapeutics, Inc. (Nasdaq: IKT) has shown in animal models of progressive disease that its lead clinical candidate, IkT-148009, is a brain penetrant Abelson tyrosine kinase, or c-Abl inhibitor, that halts disease progression and reverses functional loss in the brain and reverses neurological dysfunction in the GI tract in animal models of human disease.
The ability to halt progression and restore function was shown in animal models of progressive disease that mimic the rate of disease progression and the extent of functional loss in the brain and/or the GI tract as found in patients with PD. Small-molecule Medications with World-class Potential.
LATEST NEWS
Inhibikase Therapeutics to Present at the 11th Annual Alzheimer's & Parkinson's Drug Development Summit
IDENTIFYING THE OPPORTUNITY
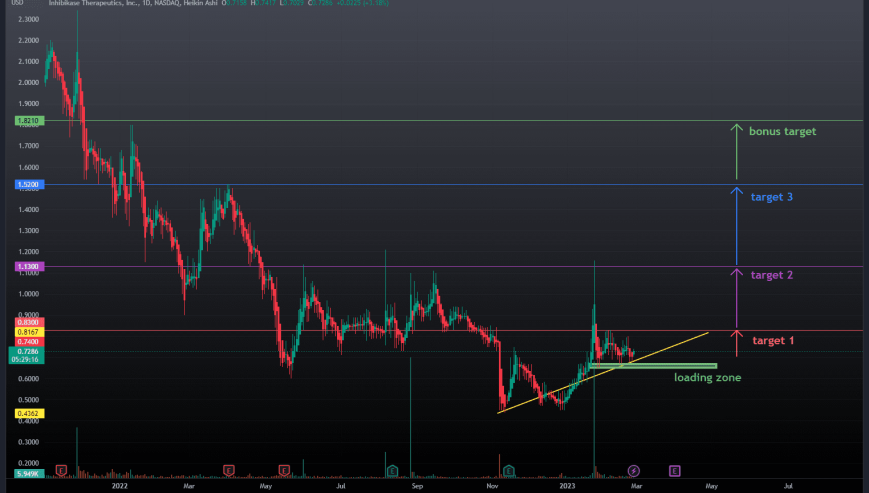
Wedge & Hedge Provides Golden Opportunity
This low-float biotech with big ratios is ready for launch!
TRADE CONFIRMATION
Wedge Consolidation setting up a strong potential for strong movement in a short time span. We are approaching a push over the 50 RSI on the daily time frame, a precursing display of breaking out. Sustained daily volume over 40k should indicate strong bullish movement. Break over .76 could set up a strong impulsive break out!
TARGETS
Day to Week: $0.83 (+17%)
1 Week: $1.13 (+60%)
1 to 2 Weeks: $1.52 (+105%)
2 Weeks+: $1.82 (+150%)
Small-molecule Medications with World-class Potential
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) used their innovative drug development platform called Re-engineering Approach with Metabolism Preserved (RAMP™) to create their initial product candidates. These candidates utilize a targeting strategy that involves blocking the activation of Abl kinases, which can be used to develop small-molecule medications capable of stopping the progression of diseases in various therapeutic areas including:

Inhibikase Therapeutics, Inc. (Nasdaq: IKT) believes its therapeutic approach would be disease-modifying.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT)’s understanding of how and why PD progresses has led the company to believe that functional loss in Parkinson’s patients may be at least partially reversed although this has not been shown clinically.
Based on the measurements in animal models, it is possible that patients treated with IkT-148009 may have their disease progression slowed or halted, the company may see a progressive reduction in the need for symptomatic or supportive therapy and/or ultimately eliminate the need for symptomatic therapy.
Reversing the Effects of Neurodegenerative Diseases
Inhibikase Therapeutics, Inc. (Nasdaq: IKT)’s therapeutics development programs focus on halting and reversing the effects of neurodegenerative diseases inside and outside of the brain. The company is also developing therapeutics that target the source of neurological infections.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT)’s pipeline includes multiple products developed from their proprietary RAMP™ dr-ug innovation and prodrug technology engines that address multiple indications.
Abl Kinases and CNS Diseases
The protein kinase family known as the Abelson Tyrosine Kinase, or Abl kinases, has been shown to play a critical role in monitoring insults to brain neurons and regulating biological pathways that are associated with neurodegeneration.
In addition, recent research has demonstrated that Abl kinases are essential checkpoint regulators that play a central role in Parkinson’s disease initiation and progression.
The breakthrough cancer treatment, imatinib (marketed as Gleevec®), was the first FDA-approved Abl kinase inhibitor.
However, imatinib cannot treat in the brain nor inhibit the wildtype enzyme in patients, even though it has a favorable safety and pharmacology profile.
Applying Inhibikase Therapeutics, Inc. (Nasdaq: IKT)’s RAMP™ technology to imatinib, Inhibikase generated novel chemical entities that have:
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) believes these molecules have the potential to be dosed at low levels with an improved safety profile relative to other drugs in this class currently used to treat certain forms of blood and gastrointestinal cancers, resulting in a safer Abl kinase inhibitor that could be chronically and systemically administered for the treatment of their CNS indications.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) Receives Favorable FDA Decision Allowing the Company to Immediately Move Forward with Phase 2a Clinical Trials.

“We are grateful for the expeditious review by the FDA of our response to the Clinical Hold on IkT-148009 in PD”
Milton H. Werner, Ph.D., President and Chief Executive Officer of Inhibikase Therapeutics, Inc. (Nasdaq: IKT)
“We believe that we now have clarity on the FDA’s expectations as we move forward in the 201 clinical trial for IkT-148009. We are now working to re-open clinical trial sites and initiate screening and enrollment of patients for the trial following agreed upon updates to the Protocol and Informed Consent form. We anticipate completing these restart tasks by the end of the first quarter.”
IkT-148009 is a c-Abl tyrosine kinase inhibitor that has been shown to halt disease progression, protect and restore lost neurons and to clear the underlying protein pathology in animal studies that suggests a causal link to the initiation and progression of disease in humans.
In lifting the clinical hold, the Agency based their decision on the Company’s Complete Response and Amendment dated December 21, 2022, as well as further commitments made on January 20, 2023 regarding ophthalmologic monitoring in the protocol of study IkT-148009-201 and various modifications to the Investigator Brochure.
The Agency requested that the Company measure the safety and steady-state pharmacokinetic (PK) profile of the 200 mg dose in six (6) healthy subjects prior to administration of the 200 mg dose in Parkinson’s patients.
The 201 trial will resume at the 50 mg and 100 mg dose immediately and the safety/PK measurement at 200 mg will be performed simultaneously.
The Agency further requested the measurement of visual acuity and examination of the cornea and lens to complement the analysis of retina, macula and fundus that was already part of the ocular monitoring program in the 201 trial.
This monitoring program is consistent with the monitoring program for ocular pathology of other approved protein kinase inhibitors. To date, no ocular pathology has been observed in any trial participant administered IkT-148009.
The Agency further requested removal of safety-related data in the Investigator Brochure for the primary metabolites of IkT-148009 to give the Agency time to review the underlying report in support of this safety data.
With agreement on conditions for restart of the 201 trial and the lifting of the clinical hold on IkT-148009, the Company intends to seek a lifting of the Clinical Hold on its program focused on Multiple System Atrophy (MSA).
Inhibikase Therapeutics, Inc. (Nasdaq: IKT): Focused on the Unmet Need in the Management of Parkinson’s Disease
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) Announces Advancement of IkT-148009 Phase 2 '201' Program Following FDA Review
On June 13, 2022, Inhibikase Therapeutics, Inc. (Nasdaq: IKT) announced that it is advancing its Phase 2a study following review of the study protocol and Phase 1/1b data by the U.S. Food and Drug Administration.
The 101 trial was a single and multiple ascending dose safety, tolerability and pharmacokinetics (PK) trial evaluating once daily administration of IkT-148009, the Company’s lead c-Abl inhibitor for the treatment of Parkinson’s disease. The study evaluated single doses up to 325 mg per day and multiple doses up to 100 mg in 88 older and elderly healthy adults and subsequently in 13 patients with mild to moderately advanced Parkinson’s disease. Clinical pharmacology of IkT-148009 in patients closely paralleled the clinical pharmacology of IkT-148009 in older healthy volunteers. IkT-148009 also demonstrated a favorable safety and tolerability profile up to a dose of 325 mg with no clinically significant adverse events observed.
Following a review of IkT-148009 safety, tolerability and PK data in the first two cohorts of the 101 trial in Parkinson’s patients at 50 and 100 mg as well as the 201 trial protocol, the FDA agreed with the Company’s view that proceeding with the 201 trial was appropriate without restrictions. Based on this feedback, Inhibikase will continue to advance the 201 trial and end enrollment of the 101 trial.
“The 101 trial was the first in human study of our novel c-Abl inhibitor, IkT-148009, which has been designed to alter the course of Parkinson’s disease. The favorable safety, tolerability and PK data of IkT-148009 observed to date supports the continued pursuit of IkT-148009 clinically,” commented Milton H. Werner, PhD, President and Chief Executive Officer.
“Based on these data and agreement with the FDA, we are very pleased to open enrollment in our 201 trial, just 17 months from the first-in-human dose of IkT-148009. This study will allow us to further evaluate the long-term safety and potential benefit of IkT-148009 in patients with Parkinson’s disease. With our first two trial sites actively screening patients, we expect to dose the first patient in the 201 trial by the end of the second quarter.”
“The 101 trial and other early studies represent key milestones that have now laid the foundation for critical trials that will define the clinical efficacy and safety of IkT-148009 over longer time periods,” said Robert A. Hauser, MD, Director of the Parkinson’s Disease and Movement Disorders Center of the University of South Florida.
The 201 trial is a 3:1 randomized, double-blind, twelve-week dosing trial that will measure the safety, tolerability and steady-state PK of IkT-148009 in untreated Parkinson’s patients as primary endpoints. The trial will also measure a hierarchy of Parkinson’s-related disease assessments in the brain and gut as secondary or exploratory endpoints. Inhibikase expects that at least 7 of 40 total sites will be open by the end of the second quarter this year.
“Parkinson’s disease is an inexorably progressive disorder and a disease-modifying therapy that slows or halts progression remains a major unmet need in the management of this disorder,” said C. Warren Olanow, M.D., FRCPC, Professor Emeritus in the Department of Neurology and Department of Neuroscience at the Mount Sinai School of Medicine in New York, and Interim Chief Medical Officer of Inhibikase Therapeutics.
“IkT-148009 has shown a good safety profile in initial SAD and MAD clinical studies as well as encouraging results in relevant humanized animal models of PD. We are excited to begin enrolling patients in our double blind placebo controlled 201 trial, which will evaluate the safety and potential disease modifying effects of IkT-148009 in a longer term setting for untreated Parkinson’s patients.“
The Global Parkinson’s Disease Treatment Market is Expected to Reach
Over $12 Billion by 2030
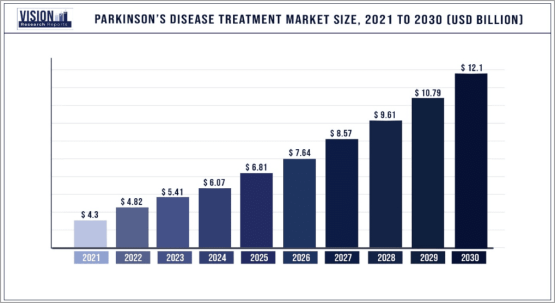
The market for treatments for neurodegenerative and infectious diseases is expected to be significant, providing an opportunity for Inhibikase Therapeutics, Inc. (Nasdaq: IKT) to capture a share of that market.
The global neurodegenerative disorder therapeutics market grew from $15.86 billion in 2022 to $17.14 billion in 2023 at a compound annual growth rate (CAGR) of 8.1%.
The market is expected to grow from $23.65 billion in 2027 at a CAGR of 8.4%.
Increased prevalence of neurodegenerative among the global population contributes to the growth of the neurodegenerative disorder therapeutics market.
The increase in the geriatric population is attributed to the increased prevalence of neurodegenerative disorders. According to World Population Prospects, the number of people above 60 years of age will increase to 2.1 billion in 2050 and it is expected that people aged over 80 years will triple to 425 million in 2050.
According to the Alzheimer’s Association, around 5.8 million Americans are suffering from Alzheimer’s disease, and this number is projected to rise to around 14 million by 2050. The increased prevalence of neurodegenerative diseases among the aging population increases the demand for neurodegenerative disorder therapeutics thereby driving the market.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) Has Established Strong Partnerships with Leading Research Institutions
Collaborations are central components of Inhibikase Therapeutics, Inc. (Nasdaq: IKT)’s strategy to build and advance our pipeline of product candidates.
Through NIH research grants awarded to Dr. Werner, we have subcontracted research projects in the biochemistry of neurodegeneration and neurological infection to Johns Hopkins University, University of Massachusetts Medical School — Worcester Campus, University of Alabama at Birmingham and the Louisiana State University Shreveport.
In oncology, Inhibikase Therapeutics, Inc. (Nasdaq: IKT) has subcontracted research work to the University of California, San Francisco and the CML Consortium and consulted with clinicians at the Memorial Sloan-Kettering Cancer Center.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT)’s research endeavors have been validated by private and public granting agencies, to include the Michael J. Fox Foundation, and the National Institute of Neurological Disease and Stroke, the National Cancer Institute and the National Institute of Allergy and Infectious Disease.
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) believes that accessing external innovation is important to their success and they plan to remain active in accessing external innovation through business development activities and awarding of private, state and federal grants through institutions such as NIH and DoD.

Inhibikase Therapeutics, Inc. (Nasdaq: IKT) Focused on Developing Novel Therapeutics Across a Wide Spectrum Including Neurodegeneration, Oncology and Infectious Diseases.
COMPANY HIGHLIGHTS
Inhibikase Therapeutics, Inc. (Nasdaq: IKT) has an experienced management team with a track record of success in drug development and commercialization.
Dr. Milton Werner is the President and Chief Executive Officer of Inhibikase Therapeutics, a company developing novel protein kinase inhibitor therapeutics to treat neurodegenerative disease and viral infections inside and outside of the brain. Previously, Dr. Werner served as Vice President of Research at Celtaxsys, a cell-free immunotherapeutics company. From September 1996 until June 2007, Dr. Werner was a Head of the Laboratory of Molecular Biophysics at The Rockefeller University in New York City. Throughout his scientific career, Dr. Werner has been an innovator integrating chemistry, physics, and biology into a comprehensive approach to solving problems in medicine, including an explanation of the origin of “maleness” in humans, the mechanistic basis of several forms of leukemia and lymphoma and, more recently, the development of therapeutics that can halt and potentially reverse functional loss in neurodegenerative disease. Dr. Werner is the author or co-author of more than 70 research articles, reviews, and book chapters and has given lectures on his research work throughout the world. He is the recipient of numerous private and public research grants totaling more than $30 million. He is the recipient of several awards, including the Naito Memorial Foundation Prize, the Young Investigator Award from the Sidney Kimmel Cancer Foundation, the Research Chair from the Brain Tumor Society, and a $1 million Distinguished Young Scholars in Medical Research Award from the W. M. Keck Foundation. Dr. Werner received his Doctor of Philosophy in Chemistry from the University of California, Berkeley, and his Bachelor of Science in Biochemistry from the University of Southern California, and he was an NIH intramural postdoctoral fellow prior to his tenure at the Rockefeller University.
Dr. C. Warren Olanow is Interim Chief Medical Officer of Inhibikase and Chief Executive Officer of CLINTREX. He is the former Henry P. and Georgette Goldschmidt Professor and Chairman of the Department of Neurology at the Mount Sinai School of Medicine in New York City, where he is presently Professor Emeritus in the Department of Neurology and in the Department of Neuroscience. Prior to joining Mount Sinai, he served on the faculties of McGill University, Duke University, and the University of South Florida. He is the former President of the Movement Disorder Society, past President of the International Society of Motor Disturbances, and former Treasurer of the American Neurological Association. He has served on the Board of Directors of the National Space Biomedical Research Institute and the executive committee of the Michael J. Fox Foundation Scientific Advisory Board, and he is the former Chairman of the Scientific Advisory Board of the Bachmann-Strauss Parkinson and of the Dystonia Foundation. Dr. Olanow is the former Co-Editor-in-Chief of the journal, Movement Disorders. He has been principal investigator of numerous studies leading to approval of drugs and devices for treating neurodegenerative diseases. Dr. Olanow received his medical degree from the University of Toronto, performed his neurology training at the New York Neurological Institute at Columbia Presbyterian Medical Center at Columbia University, and undertook postgraduate studies in neuroanatomy at Columbia University.
Joseph Frattaroli has served as our Chief Financial Officer since April 2018. Mr. Frattaroli is a certified public accountant with more than 15 years of experience in public company filings and compliance for Nasdaq and OTC Markets companies. He founded Flagship Consulting, Inc. in January 2010, through which he has provided chief financial officer and consulting services for several emerging biopharmaceutical and medical device companies, with responsibilities that included capital formation, deal structuring, and assisting private companies in their transition to becoming publicly traded SEC registrants. He has also served as an independent consultant to Danforth Advisors from July 2015 until joining Inhibikase, providing interim chief financial officer and strategic advisory services to emerging public and private biotechnology and biopharmaceutical clients. Mr. Frattaroli received his Bachelor of Science in Accounting from Salem State University and was certified as a public accountant while employed by Ernst & Young, LLP. Mr. Nijjar holds a CPA CMA designation from the Chartered Professional Accountants of British Columbia and a BComm from the University of British Columbia. Other Issuers: CFO for Playgon Games Inc., Marvel Biosciences Corp., Los Andes Copper Ltd., Forty Pillars Mining Corp.
Dan Williams acts as the Company’s controller through his own company Startup Personnel, LLC
Dr. Terence Kelly is a 30-year pharmaceutical industry veteran and has served as a member of the board of directors of Cardax since June 2014. He is a founder of Kelly Pharma Research Consulting, LLC, and has served as its President since January 2010. From June 2010 to July 2017, he held several positions at CoMentis, including most recently President and CEO. From July 2002 to December 2009, he served as Vice President of Medicinal Chemistry at Boehringer Ingelheim Pharmaceuticals, where he, developed processes for the RAMP drug discovery program. Mr. Wong currently sits on the board of directors of Plant Veda Foods Ltd. (CSE: MILK).
Dr. Roger Rush is an experienced veteran of the pharmaceutical industry with more than 30 years of experience working in the United Kingdom and the U.S. for small and large pharmaceutical companies and contract research organizations and is now based in the Greater Boston area. His major career focus has been on preclinical research and development, safety assessment, and the translation of discovery research molecules into clinical development. He has contributed to more than 20 IND, CTA, and product license submissions and approved drugs, including nicardipine (Cardene), ranolazine (Ranexa), Foscan, and zileuton (Zyflo CR). He has been a principal of Allon Preclinical Consulting, LLC since 2015. From March 2012 to December 2014, he was Vice President, Preclinical Development for Idenix Pharmaceuticals, Inc., a wholly owned subsidiary of Merck & Company, Inc., where he managed the DMPK, toxicology, and discovery research that led to the identification of lead molecules to treat Hepatitis C virus. His work has spanned numerous therapeutic areas, including anti-inflammatory, anti-allergy, arthritis, anti-infectives, CNS, cardiovascular, oncology, genitourinary, and anti- antihyperlipidemic agents. He received his Bachelor of Science and Doctor of Philosophy in Biochemistry from the University of Surrey in the United Kingdom.
Dr. Surendra Singh is an expert in chemical process research and development from lead optimization to launch, technology transfer, and API manufacturing. From 2001 to 2011, he served various roles at Sunovion Pharmaceuticals Inc. and its predecessor, Sepracor Inc., including as a director of chemical process research, and has served as a chemistry, manufacturing, and control consultant for AVEO Oncology, RestorGenex Corporation, Verastem Oncology, and Syner-G Pharma Consulting, LLC. Dr. Singh received his doctoral degree from the Indian Institute of Technology in 1991 and was a postdoctoral fellow at The Ohio State University. Dr. Singh establishes and manages the commercial process, global outsourcing, and global vendor management as well as participates in all aspects of the drafting and review of regulatory documents, from IND to NDA.
Merrill M. Kraines is a partner in the Health Sciences practice of McDermott, Will and Emery LLP, resident in the New York office. Mr. Kraines is primarily engaged in corporate, securities, and mergers and acquisitions matters. He leads the life sciences, technology, and emerging growth practices in New York. Mr. Kraines represents many life sciences companies, including biotechnology, therapeutic, diagnostic, and medical device companies. He has also represented or been involved in transactions concerning a variety of other growth companies in all phases of business and finance in many industries, including software, Internet, health care, IT, alternative energy, financial services, IT consulting, educational publishing, entertainment, office products, advertising, telecommunications, paper, and many other companies. These companies have ranged from start-ups to large public concerns.
Voisin Life Science Consulting is a regulatory affairs consultancy with extensive regulatory experience, representing pharmaceutical and biotechnology companies at the U.S. FDA and EMA. The company has helped to guide development programs for small molecules and biologics through all phases of regulatory review.
Sources
- Source 1: https://finance.yahoo.com/news/inhibikase-therapeutics-announces-fda-lifted-130500546.html
- Source 2: https://www.biospace.com/article/parkinson-s-disease-treatment-market-size-share-report-2022-2030/
- Source 3: https://www.inhibikase.com/investors/sec-filings/all-sec-filings/content/0000950170-22-005203/0000950170-22-005203.pdf
- Source 4: https://www.sec.gov/Archives/edgar/data/1750149/000095012321007495/filename1.htm
- Source 5: https://www.inhibikase.com/news/press-releases/detail/63/inhibikase-therapeutics-to-present-data-from-phase-11b
- Source 6: https://www.prnewswire.com/news-releases/inhibikase-therapeutics-announces-fda-has-lifted-the-full-clinical-hold-on-ikt-148009-in-parkinsons-disease-301729466.html
- Source 7: https://fintel.io/so/us/ikt
- Source 8: https://www.investopedia.com/terms/n/nanocap.asp
- Source 9: https://www.benzinga.com/money/best-nano-cap-stocks
- Source 10: https://www.marketwatch.com/investing/stock/ikt?mod=search_symbol
- Source 11: https://www.barchart.com/stocks/quotes/IKT/price-history/historical?orderBy=highPrice&orderDir=desc
- Source 12: https://stockstotrade.com/low-float-stocks/
- Source 13: https://www.sofi.com/learn/content/understanding-low-float-stocks/
- Source 14: https://finviz.com/quote.ashx?t=IKT&p=d
- Source 15: https://www.investopedia.com/articles/stocks/05/042605.asp
- Source 16: https://schrts.co/VWdBYwMe
- Source 17: https://d1io3yog0oux5.cloudfront.net/_023374ccb372c17bdbdd673bc93fa94b/inhibikase/files/theme/images/logo-sm.svg
- Source 18: https://www.raps.org/RAPS/media/news-images/news-images/FDA-Logo.jpg
- Source 19: https://www.fiercebiotech.com/biotech/inhibikase-stock-sinks-fdas-unexpected-clinical-hold-lead-assets-parkinsons-trial
- Source 20: https://ars.els-cdn.com/content/image/1-s2.0-S0165614715000772-gr1.jpg
- Source 21: https://www.inhibikase.com/technology/ramp-drug-innovation-engine
- Source 22: https://d1io3yog0oux5.cloudfront.net/_023374ccb372c17bdbdd673bc93fa94b/inhibikase/db/998/8459/image.jpg
- Source 23: https://m3.healio.com/~/media/slack-news/infectious-disease/misc/other/fda_building.jpg
- Source 24: https://d1io3yog0oux5.cloudfront.net/_023374ccb372c17bdbdd673bc93fa94b/inhibikase/db/935/8710/pdf/IKT+2022+Presentation+-1Q23v2.pdf
- Source 25: https://www.inhibikase.com/news/press-releases/detail/56/inhibikase-therapeutics-announces-advancement-of-ikt-148009
- Source 26: https://assets.technologynetworks.com/production/dynamic/images/content/369307/promising-parkinsons-disease-treatment-candidate-identified-in-mouse-study-369307-960×540.jpg?cb=12124001
- Source 27: https://www.biospace.com/article/parkinson-s-disease-treatment-market-size-share-report-2022-2030/
- Source 28: https://www.thebusinessresearchcompany.com/report/neurodegenerative-disorder-therapeutic-global-market-report
- Source 29: https://www.inhibikase.com/about/management-team
- Source 30: https://i.guim.co.uk/img/media/bb3e9fe9deab523c9c20a066db3634c21f20a155/415_84_3886_3086/master/3886.jpg
- Source 31: https://exxelpharma.com/wp-content/uploads/2018/01/7453878A177E47EF8C214AB7EBFB7631.png
Disclaimer
Stock Research Today is a project of Virtus Media Group LLC and intended solely for entertainment and informational purposes. Consult your financial, investment and tax advisors to determine what financial and tax strategies may be right for you. Investor protection and other important information is available at https://www.sec.gov/. This website / media webpage is owned, operated and edited by Virtus Media LLC. Any wording found on this website / media webpage or disclaimer referencing “I” or “we” or “our” or “Virtus Media” refers to Virtus Media LLC. This website / media webpage is a paid advertisement, not a recommendation nor an offer to buy or sell securities. Our business model is to be financially compensated to market and promote small public companies. By reading our website / media webpage you agree to the terms of our disclaimer, which are subject to change at any time. We are not registered or licensed in any jurisdiction whatsoever to provide investing advice or anything of an advisory or consultancy nature and therefore are unqualified to give investment recommendations. Always do your own research and consult with a licensed investment professional before investing. This communication is never to be used as the basis for making investment decisions and is for entertainment purposes only. At most, this communication should serve only as a starting point to do your own research and consult with a licensed professional regarding the companies profiled and discussed. Conduct your own research. Companies with low price per share are speculative and carry a high degree of risk, so only invest what you can afford to lose. By using our service you agree not to hold our site, its editor’s, owners, or staff liable for any damages, financial or otherwise, that may occur due to any action you may take based on the information contained within our website / media webpage .We do not advise any reader take any specific action. Losses can be larger than expected if the company experiences any problems with liquidity or wide spreads. Our website / media webpage are for entertainment purposes only. Never invest purely based on our alerts. Gains mentioned in our website / media webpage may be based on end-of-day or intraday data. This publication and their owners and affiliates may hold positions in the securities mentioned in our alerts, which we may sell at any time without notice to our subscribers, which may have a negative impact on share prices. If we own any shares we will list the information relevant to the stock and number of shares here. Virtus Media business model is to receive financial compensation to promote public companies. To conduct investor relations advertising, marketing and publicly disseminate information not limited to our websites, email, sms, push notifications, influencers, social media postings, ticker tags, press releases, online interviews, podcasts, videos, audio ads, banner ads, native ads, responsive ads. This compensation is a major conflict of interest in our ability to be unbiased regarding. Therefore, this communication should be viewed as a commercial advertisement only. We have not investigated the background of the hiring third party or parties. The third party, profiled company, or their affiliates likely wish to liquidate shares of the profiled company at or near the time you receive this communication, which has the potential to hurt share prices. Any non-compensated alerts are purely for the purpose of expanding our database for the benefit of our future financially compensated investor relations efforts. Frequently companies profiled in our alerts may experience a large increase in volume and share price during the course of investor relations marketing, which may end as soon as the investor relations marketing ceases. Our emails may contain forward-looking statements, which are not guaranteed to materialize due to a variety of factors.
We do not guarantee the timeliness, accuracy, or completeness of the information on our website / media webpage. The information in our website / media webpage is believed to be accurate and correct, but has not been independently verified and is not guaranteed to be correct. The information is collected from public sources, such as the profiled company's website and press releases, but is not researched or verified in any way whatsoever to ensure the publicly available information is correct. Furthermore, Virtus Media often employs independent contractor writers who may make errors when researching information and preparing these communications regarding profiled companies. Independent writers' works are double-checked and verified before publication, but it is certainly possible for errors or omissions to take place during editing of independent contractor writer's communications regarding the profiled company(s). You should assume all information in all of our communications in incorrect until you personally verify the information, and again are encouraged to never invest based on the information contained in our written communications. The information in our disclaimers is subject to change at any time without notice. Compensation: Pursuant to an agreement between Virtus Media LLC and Lifewater media LLC, Virtus Media has been hired for a period beginning on 2023-02-28 and ending after 2023-03-06 to publicly disseminate information about NASDAQ: IKT. We have been paid fifteen thousand dollars USD via ACH Bank Transfer. Pursuant to an agreement between Virtus Media LLC and Lifewater media LLC, Virtus Media has been hired for a period beginning on 2023-03-08 and ending after 2023-03-13 to publicly disseminate information about NASDAQ: IKT. We have been paid fifteen thousand dollars USD via ACH Bank Transfer.
